Clinical features
Two years prior to admission, the patient developed sudden-onset of intermittent palpitations, which he initially tolerated, and for which he did not seek medical consultation. One month before admission, however, his symptoms worsened, and became associated with occasional chest discomfort and two-pillow orthopnea. This prompted him to consult a cardiologist, who advised 2D-echocardiogram. Imaging revealed an atrial septal defect (ASD) with concomitant mitral stenosis (MS), consistent with Lutembacher syndrome (LS). He was subsequently referred to our service for surgical management.The patient initially denied any history of tonsillopharyngitis or rheumatic heart disease (RHD). He did not smoke, drink alcohol or use illicit drugs. There was no family history of congenital heart disease. He denied any episodes of syncope, cyanosis, or exertional dyspnea. On chest examination, a parasternal heave was appreciated on palpation. Cardiac rate and rhythm were regular. Auscultation revealed a grade 4/6 holosystolic murmur at the left 2nd to 4th intercostal spaces, parasternal to midclavicular line, radiating to the mid-axillary area. There was no neck vein engorgement, hepatosplenomegaly, or bipedal edema. The rest of the physical examination findings were unremarkable.
Diagnostics
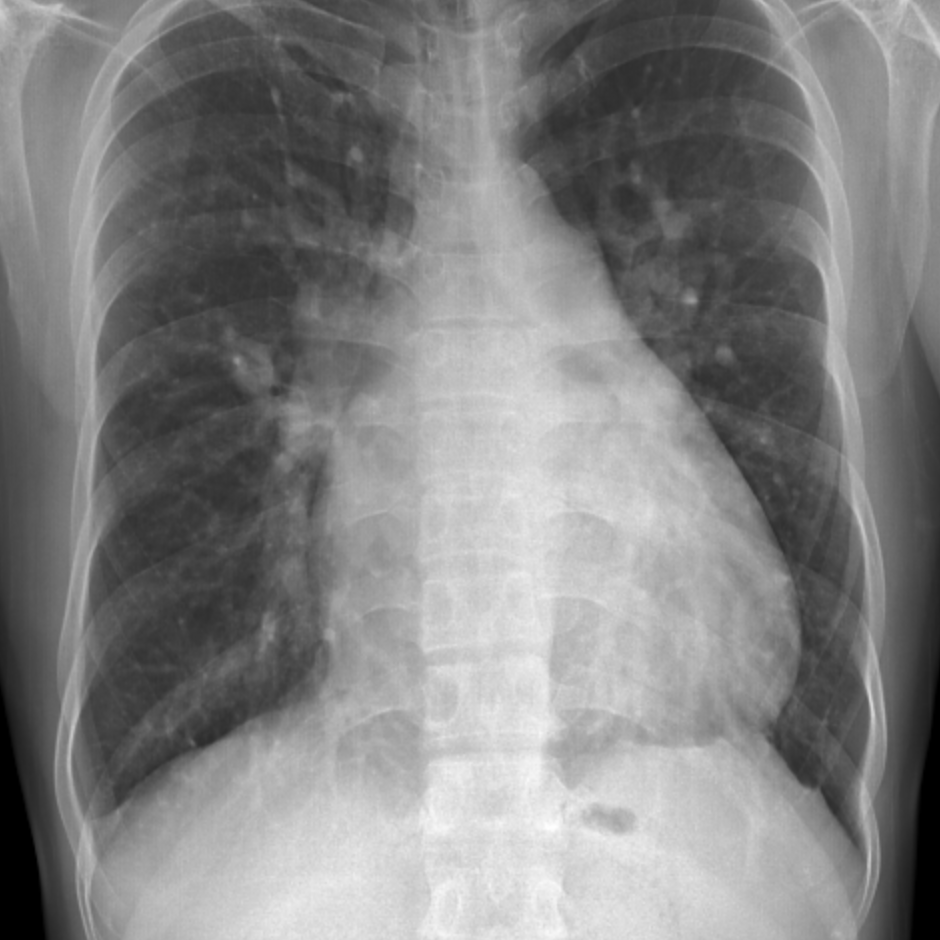
A chest radiograph revealed gross cardiomegaly, pulmonary congestion, and minimal right pleural effusion (Figure 1). Preoperative 2D echocardiography showed a normal left ventricular dimension with normal wall motion and contractility, and a left ventricular ejection fraction of 63%. The right atrium, left atrium, and right ventricle were all dilated. There was a moderate MS, with mitral valve area of 1.5 cm2 by both planimetry and pressure half-time, and mild mitral regurgitation. A concomitant secundum-type ASD was noted, with predominant left-to-right shunting and a Qp/Qs ratio >1.5:1. Mild pulmonic hypertension was present, with a pulmonary artery pressure of 42 mmHg. Histopathology of the excised mitral valve revealed areas of fibrosis.Therapeutics
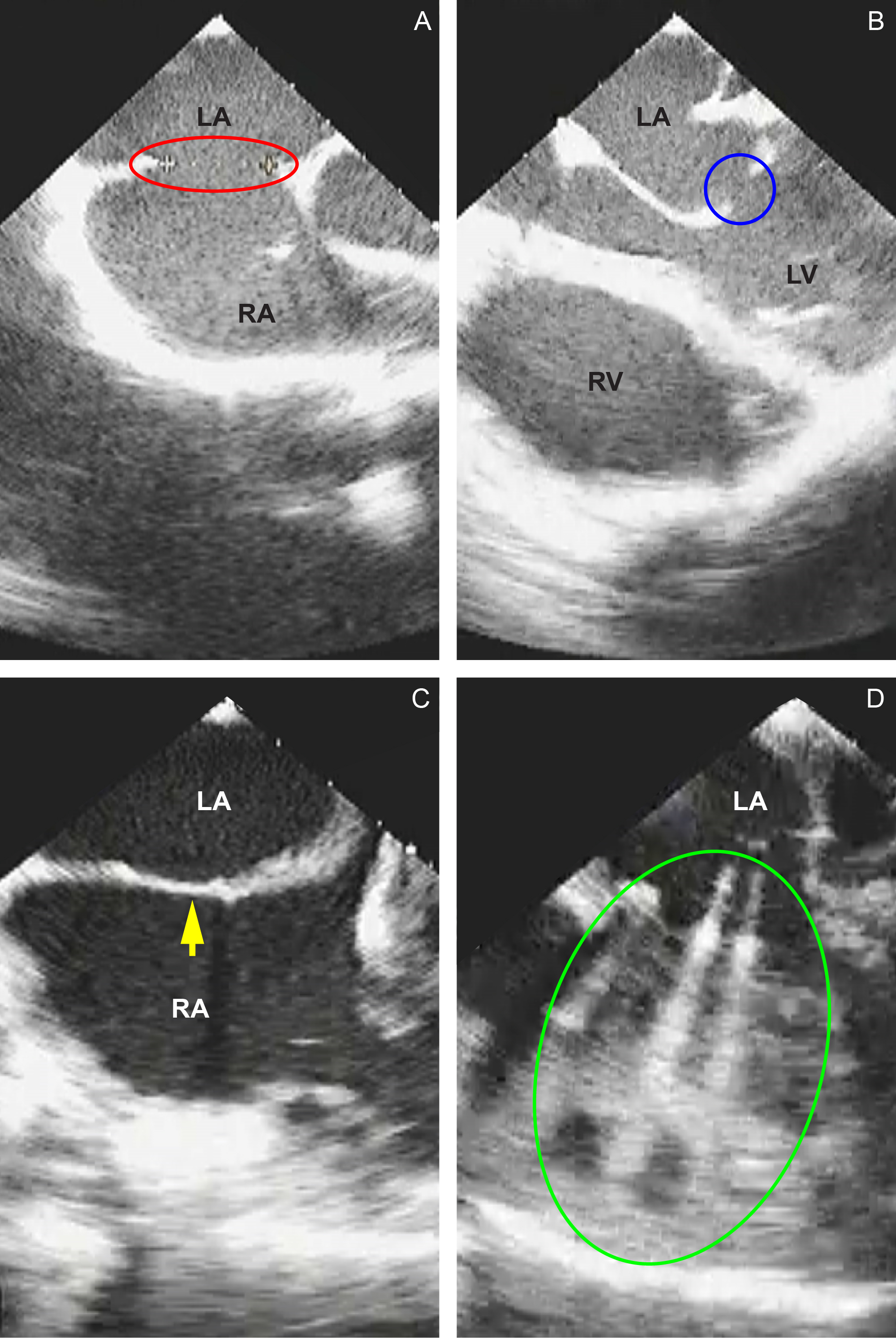
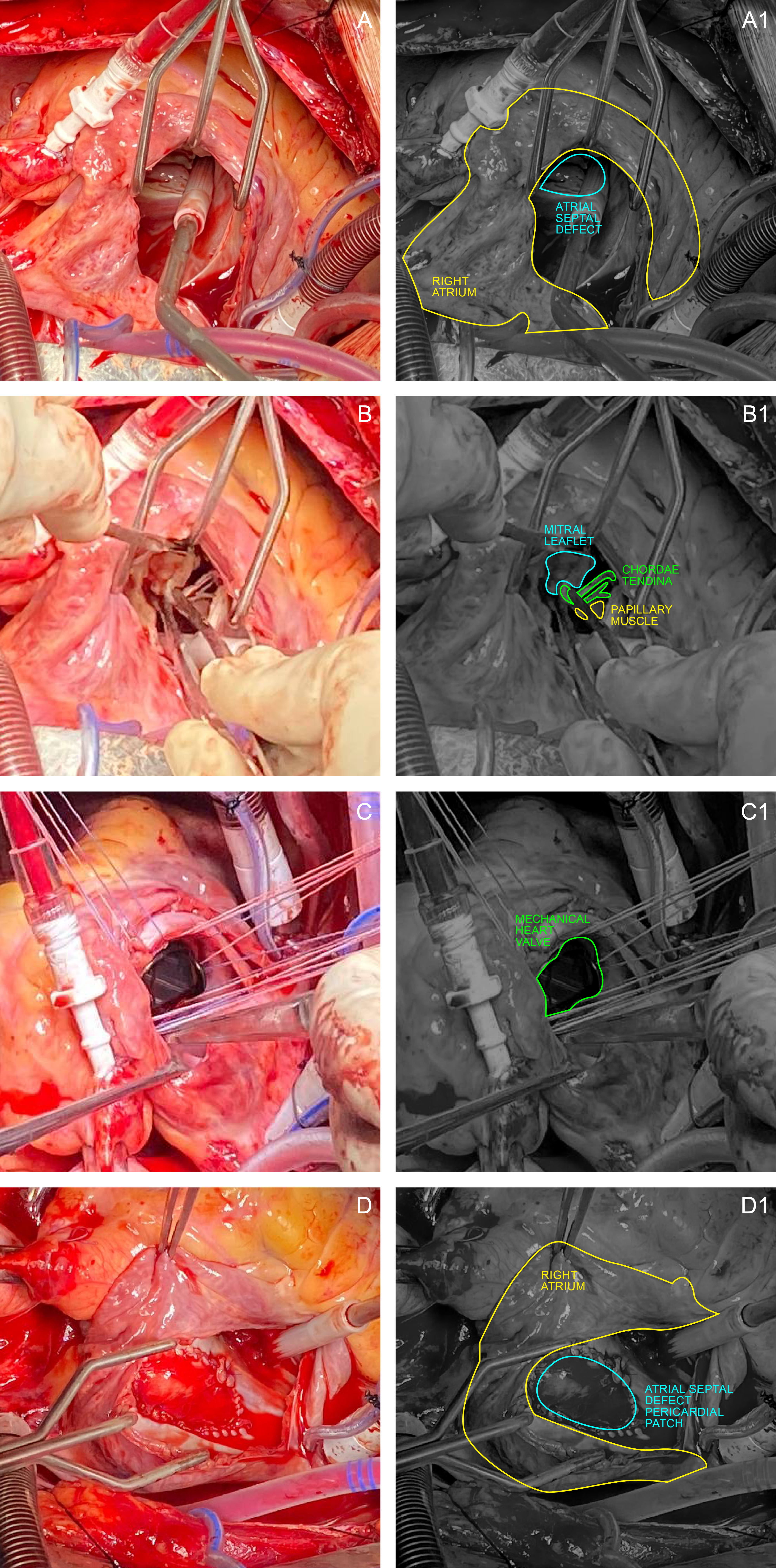
The patient was admitted to our institution and comanaged with the Cardiology Service, which performed an intraoperative transesophageal echocardiogram (TEE) (Figure 2A and 2B) prior to the mitral valve replacement and patch closure of the ASD. We carried out the procedure via a median sternotomy. After achieving adequate anticoagulation, aortic and bicaval cannulation were performed. We initiated cardiopulmonary bypass (CPB), cooled down the patient, and applied the aortic cross-clamp. Adequate myocardial preservation was achieved using modified Del Nido cardioplegia, delivered antegrade into the aortic root, and retrograde through the coronary sinus. We then performed a right atriotomy, gaining access to the ASD (Figure 3A). We examined and excised the mitral valve through a transseptal approach (Figure 3B) and then implanted a mechanical valve (Figure 3C). Finally, we repaired the ASD using an autologous pericardial patch (Figure 3D). Post-bypass intraoperative TEE demonstrated an intact ASD patch and good function of the mitral valve prosthesis (Figure 2C and 2D). We successfully weaned the patient from CPB without any intraoperative complications.We extubated the patient on the first postoperative day. We started warfarin therapy and later removed his chest drains. By postoperative day 11, we achieved the target anticoagulation, with an INR of 2.64. We discharged him on postoperative day 12 after an excellent recovery. During follow-up, a postoperative echocardiogram revealed an intact mitral valve prosthesis with good opening and closing motion and no residual shunt at the ASD patch. The Cardiology Service closely monitored his prothrombin time—INR while he continued oral warfarin. He remained asymptomatic throughout his outpatient follow-up.
Relevance
Lutembacher’s Syndrome is a rare structural cardiac abnormality characterized by the coexistence of a congenital or iatrogenic ASD and a congenital or acquired MS.1 Congenital ASD is present in 0.6% to 0.7% of patients with MS.2 The incidence of LS in the United States has been reported at 0.001 per million people.3 The condition has been reported more frequently in Southeast Asia, where RHD remains prevalent. It is more common in females than in males. In our patient’s case, the clinical history and physical examination, echocardiographic findings, and histopathology results suggest RHD as the probable cause of the MS, despite the absence of a reported history of tonsillopharyngitis. The findings of pulmonary congestion and pleural effusion on the chest radiograph indicate possible left-sided heart failure in our patient, which may be producing the holosystolic murmur noted on physical examination. Other etiologies of LS—such as congenital mitral valve anomalies, autoimmune disease (e.g., systemic lupus erythematosus or rheumatoid arthritis), or mitral annular calcification typically associated with older age—are less likely.4The coexistence of ASD and MS results in a distinct hemodynamic interaction that significantly alters the clinical presentation compared to having either condition alone.2 5 In LS, the MS increases the magnitude of left-to-right shunting across the ASD, exacerbating right ventricular volume overload, and predisposing patients to atrial fibrillation.2 6 Our patient had moderate MS with a secundum-type ASD, making surgical intervention essential to prevent progressive pulmonary hypertension caused by increased right-sided flow. If pulmonary artery pressures continue to rise undetected, patients may eventually develop a right-to-left shunt with worsening symptoms.7
Management options that offer a favorable prognosis include surgical ASD closure with mitral valve replacement, both of which were successfully carried out on our patient. Percutaneous interventions—such as percutaneous transluminal mitral commissurotomy and ASD device closure using the Amplatzer device—may be appropriate in situations where the mitral valve and atrial septum have favorable anatomical characteristics and the ASD is hemodynamically significant.8 9 10 Ultimately, timely diagnosis and appropriate intervention are critical to prevent irreversible hemodynamic deterioration and to achieve favorable long-term outcomes for patients with Lutembacher syndrome.