Clinical features
The lesions appeared three days after the patient began taking an herbal capsule (one capsule thrice a day) containing 23 botanical extracts, including aloe vera, Siberian ginseng, licorice root, astragalus root, reishi mushroom, Chinese pearl barley, schisandra berry, rose hip, chicory root, dandelion root, German chamomile, alfalfa herb, cascara bark, fenugreek seed, bee pollen, pipsissewa, juniper berry, ginger root, celery seed, sarsaparilla, passion flower, thyme, and capsicum fruit. The patient initially reported fever, pruritus, hot flashes, and a burning skin sensation, followed by generalized erythematous patches with dusky centers. These rapidly progressed to vesicles, beginning on the lower extremities. Despite discontinuing the herbal medication, new vesicles still emerged, coalescing into large erythematous flaccid bullae on the anterior and posterior trunk. The patient also developed multiple aphthous ulcers in the oral cavity, causing dysphagia even with liquids. Lesions on the genitalia, also caused dysuria. After desquamation, the posterior trunk lesions resulted in large eroded and denuded plaques.The patient was newly diagnosed with invasive cervical carcinoma when she started the herbal capsule. She had a history of hypertension and poor compliance with amlodipine but had no history of chronic skin conditions, diabetes, peripheral vascular disease, or systemic autoimmune diseases or a family history of Stevens-Johnson Syndrome (SJS) or Toxic Epidermal Necrolysis (TEN). She denied taking other medications or dietary supplements, or applying topical products and reported no recent fever, cough, rhinitis, headache, or arthralgia.
Diagnostics and therapeutics
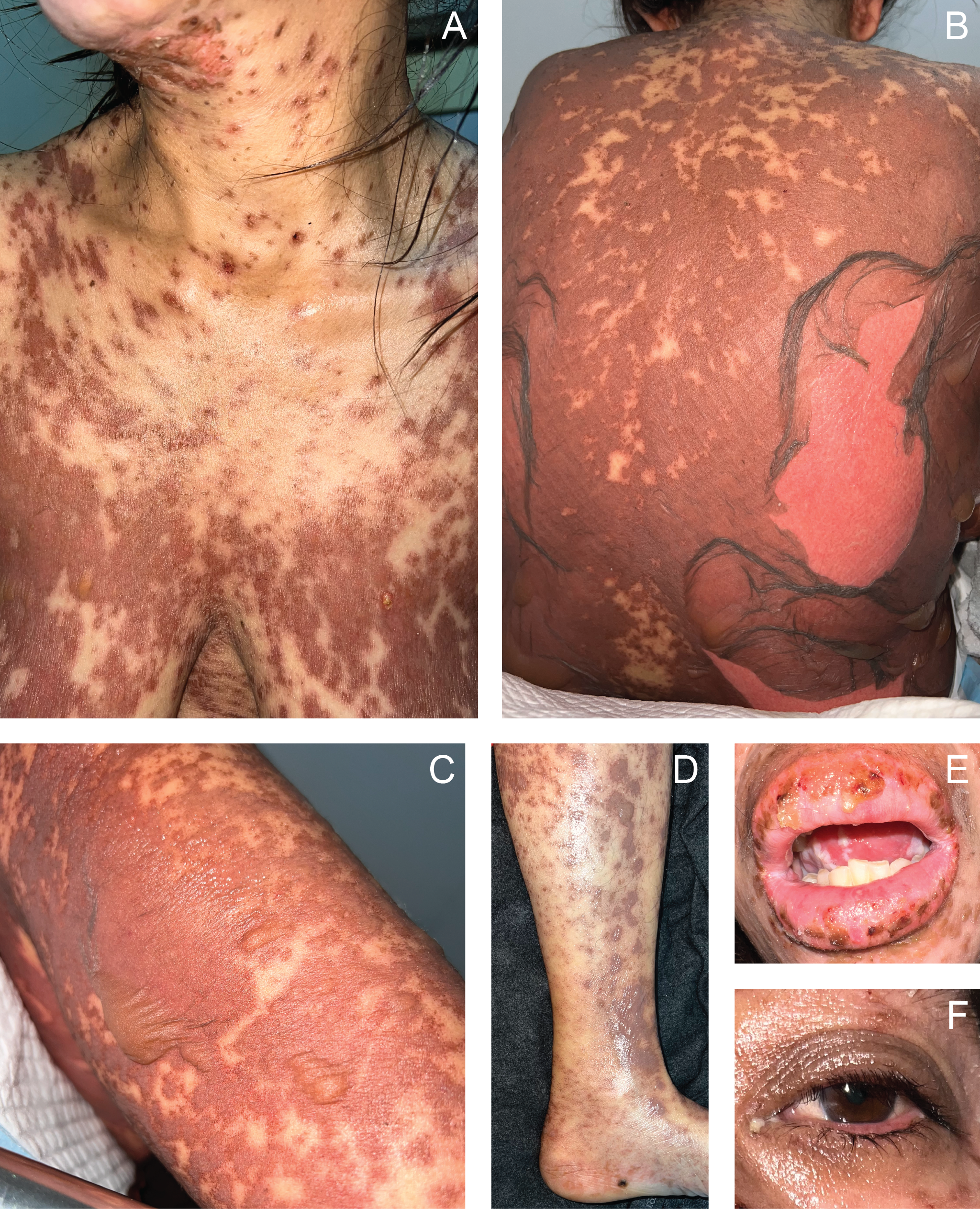
The patient was admitted to the intensive care unit (ICU) under the Internal Medicine (IM) Service and was immediately referred to our Dermatology Service for comanagement. On physical examination, we observed generalized vesicles and bullae on an erythematous background. We noted numerous fused vesicles and bullae, progressing to desquamated plaques involving the trunk (Figure 1A-B) and extremities (Figure 1C-D). Epidermal detachment involved an estimated 90% of the total body surface area (TBSA). Both Nikolsky and Asboe-Hansen signs were positive. Additionally, the patient exhibited skin tenderness, mucositis, and ulceration in the oral cavity and conjunctiva (Figure 1E-F).Based on the history and physical findings, we made an initial diagnosis of TEN. The likelihood of the herbal medication causing the condition was assessed using the Naranjo Adverse Drug Reaction Probability Scale,1 which yielded a score of 6, indicating a ‘probable’ adverse drug reaction. This suggested a reasonable temporal relationship between the administration of the herbal medication and the onset of TEN. Differential diagnoses included SJS, staphylococcal scalded skin syndrome, pemphigus vulgaris, bullous pemphigoid, erythema multiforme major, and bullous systemic lupus erythematosus. SJS, which is in the same disease spectrum as TEN, was ruled out because the TBSA affected by epidermal detachment exceeded 30%, whereas SJS involves less than 10%.2
The Severity-of-Illness Score for Toxic Epidermal Necrolysis (SCORTEN)—which considers factors such as age, presence of malignancy, and extent of epidermal detachment to assess the severity of illness and prognostic score for patients with TEN3—produced a score of 3 for this patient. This corresponded to a predicted mortality rate of 35.8%. Chest radiography was unremarkable, while laboratory results showed mild anemia, a slightly elevated leukocyte count, and mild hypoalbuminemia (27 g/L). Wound, blood, and urine cultures were negative on admission.
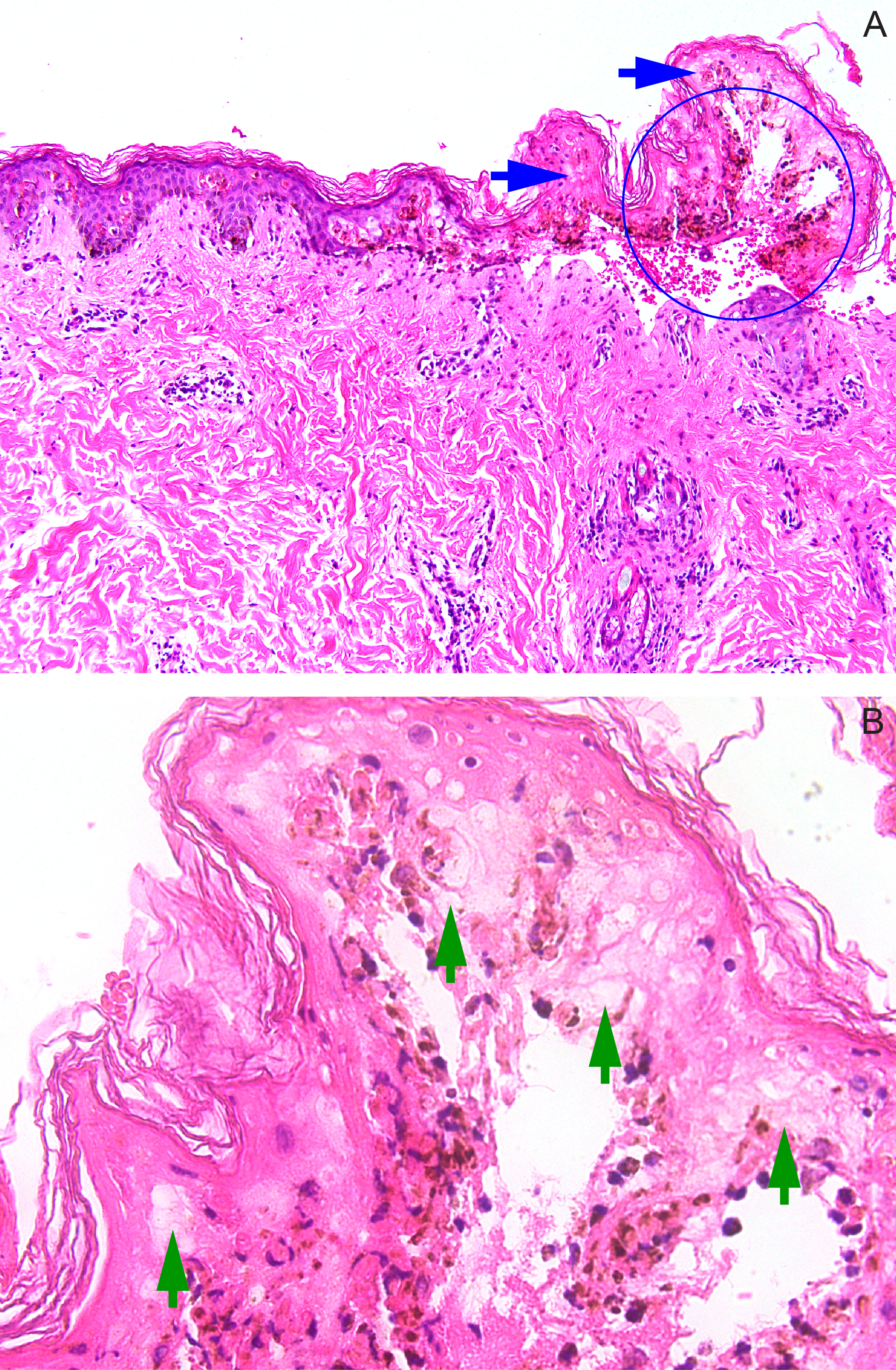
Histopathology from a skin punch biopsy revealed an atrophic epidermis with full-thickness necrosis and separation of the epidermis from the dermis, producing a subepidermal split (Figure 2A). The dermis exhibited superficial edema and mild interstitial and perivascular inflammatory infiltrates composed of lymphocytes, histiocytes, and eosinophils. Closer magnification of the blister roof revealed confluent keratinocyte necrosis and minimal inflammatory infiltrates within the blister cavity (Figure 2B). These histopathologic findings confirmed the diagnosis of TEN.
The management of TEN in our patient required a multidisciplinary approach. On admission, supportive treatment, including fluid resuscitation, pain management, and nasogastric nutritional support, was initiated by the IM Service while she was placed under intensive care. The patient was started on oral prednisone at 1 mg/kg/day, with tapering after seven days. On the third hospital day, purulent discharge from the desquamated plaques prompted a repeat wound culture revealing Staphylococcus aureus. Intravenous clindamycin was initiated, along with mupirocin ointment for the eroded areas, bilastine for the pruritus, and chlorhexidine oral gargle for hygiene. We also advised the patient on careful thermoregulation and regular application of petrolatum on the skin and lips.
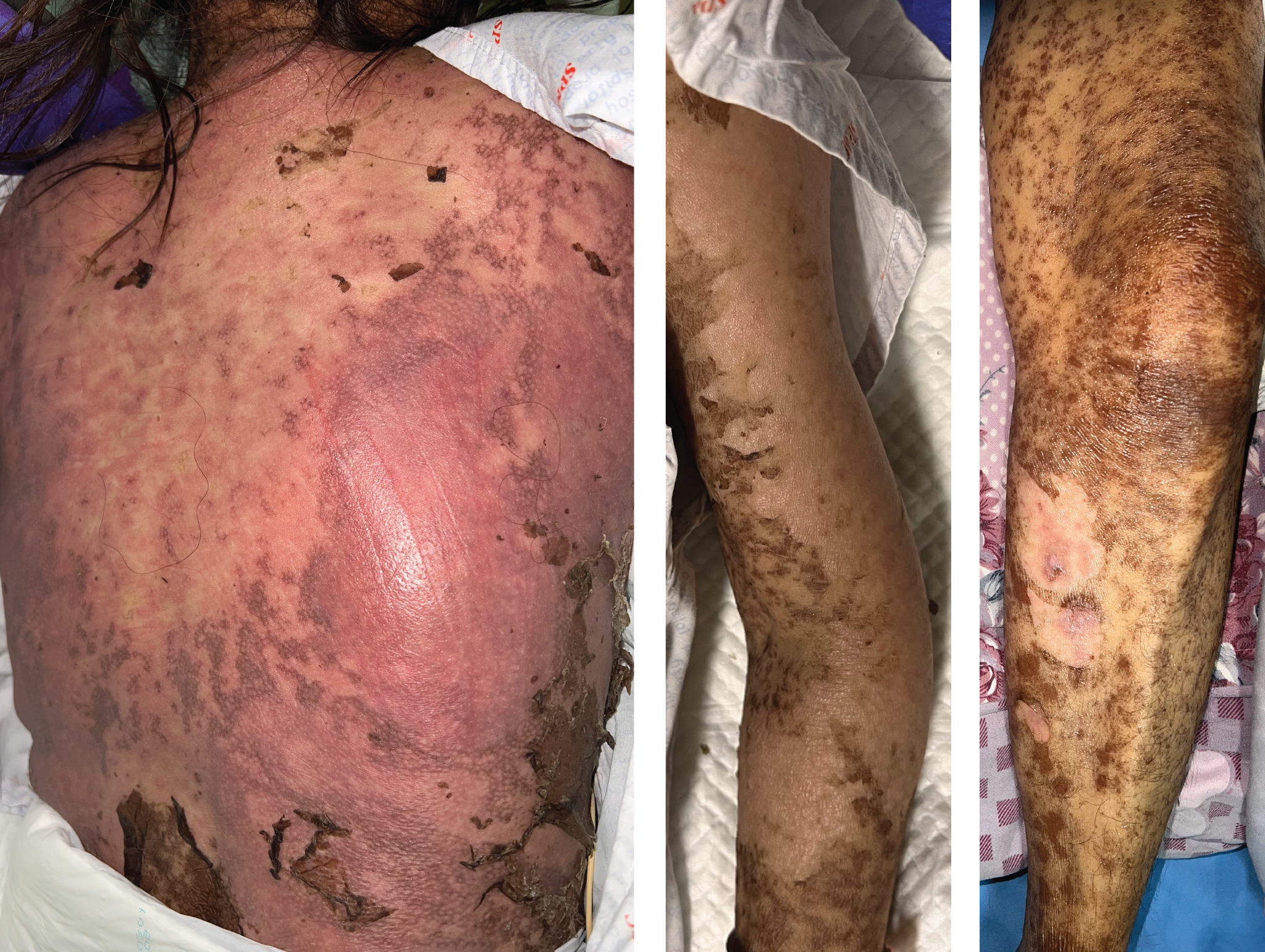
The Ophthalmology Service managed the ocular involvement with ophthalmic steroids containing anti-infectives, and advised lid hygiene. The Gynecology Service managed the patient’s cervical cancer, postponing interventions until postdischarge. On the 10th hospital day, the patient had a catheter-associated urinary tract infection, and Proteus mirabilis was identified on urine culture. She was subsequently started on intravenous ceftriaxone. By the 14th hospital day, we observed complete re-epithelialization with some areas of postinflammatory hyperpigmentation (Figure 3).
Upon discharge on the 22nd hospital day, we instructed the patient to continue taking oral prednisone for another week, along with mupirocin ointment, bilastine, ophthalmic steroids/anti-infectives, and emollients. We also added vitamin D, calcium, and iron supplements to the regimen. Our team provided wound care instructions and educated the family on signs of infections, pain management, and comfort measures. The patient followed-up at our clinic via teledermatology at one and two weeks postdischarge, where she was determined to have made a full recovery from TEN. Unfortunately, two months later, she died due to the rapid progression of her cervical squamous cell carcinoma.